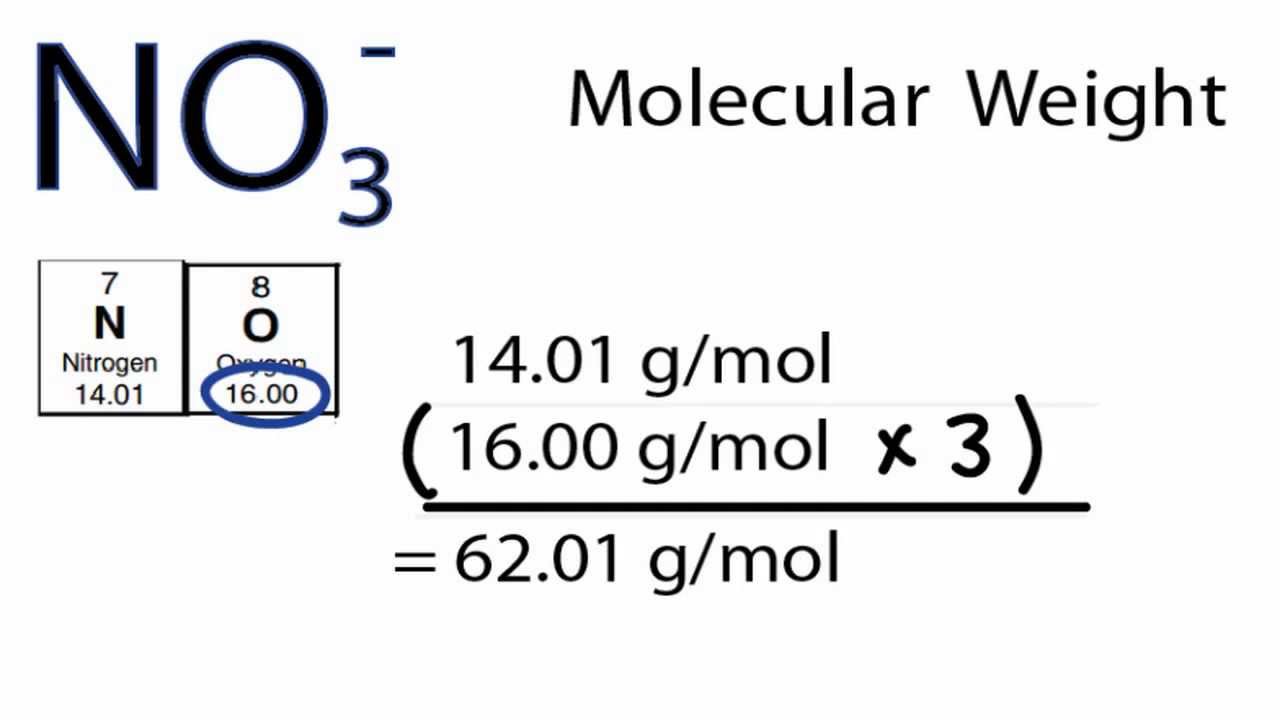 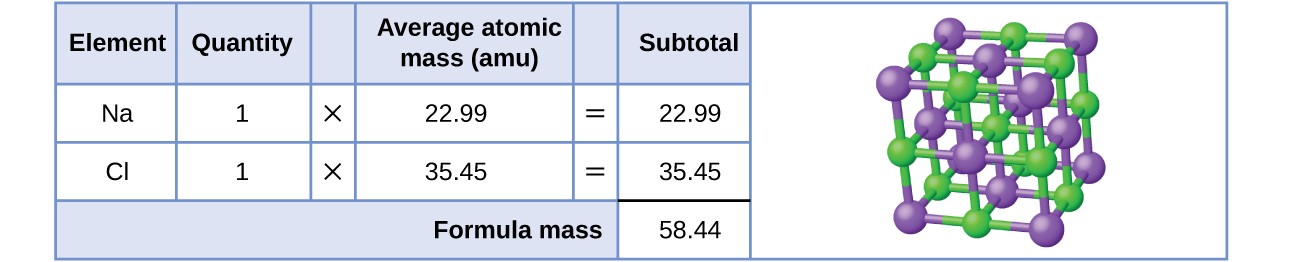
Summary Sodium Chloride vs Potassium Chloride Potassium chloride is KCl and sodium chloride is NaCl. So here we would take a look at our periodic table and we would take a look at the, uh, weighted average mass of each element sodium and quarry. Apart from that, people who don’t want to intake Na can have KCl salt, instead of NaCl table salt. If they are next to each other, multiply them to get the answer, if one is on top of the other, divide the top one by the bottom one to get the answer. Molar mass of KCl is higher than that of NaCl’ the molar mass of sodium chloride is 58.44 g/mol, and for potassium chloride, it is 74.55 g/mol. To get the formula from the triangle, cover up the value you want to calculate and use the remaining two values. At room temperature and pressure, this compound appears as solid, colorless crystals. To remember the formulas in this section, you can use the formula triangles. Sodium chloride is NaCl that has a molar mass of 58.44 g/mol. For example, about 98.9% of naturally occurring carbon is 12C ('Carbon 12') and about 1.1% is 13C so carbon has an atomic weight of \begin Formula Triangles The atomic weight (or atomic mass) of an element tells us on average how much one atom of a given element weighs, taking into account typical proportions of isotopes. of argan oil is argania spinosa kernel oil, or the INCI name of table salt (yes, it is a common ingredient in cosmetic products) is sodium chloride. M or Molarity means amount of substance concentration in 1 litre. Cacl2 industrial grade calcium chloride 94 white granular calcium chloride 1. Contents Toggle Main Menu 1 Atomic Weight 2 Molecular Weight 3 Moles 4 Solutions 4.1 1) Molarity 4.2 2) Mass Concentration 4.3 A note on units for concentrations 4.4 Diluting Solutions 5 Test yourself Atomic Weight Sodium chloride molar mass is 58.44 g mol-1 You need to understand the concept first. Molar Mass Chloride (5 products available) 1/6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
